Overview
The He Lab is dedicated to advancing Artificial Intelligence (AI) in medical image analysis for early diagnosis / prediction of various important clinical outcomes. Our research has been supported by multiple grants from the National Institute of Health and charitable foundations. We are focused on developing MRI prognostic biomarkers and deep learning models for early detection/ prediction of various important clinical outcomes, including cognitive, language, and motor deficits, attention deficit hyperactivity disorder, autism spectrum disorder, and chronic liver diseases. Our research involves the development of AI techniques for medical image classification, regression, segmentation, registration, and synthesis.
Early Prediction of Neurodevelopmental Deficits in Very Preterm Infants
Of all U.S. births (~3.79 million in 2018), 11% occur preterm. Preterm infants have an increased risk of developing neurodevelopmental deficits. The burden of preterm birth is not only shouldered by the individual children themselves, but also by their parents/families, and society as a whole. The societal economic impact of lifetime care for people born with cognitive deficits is estimated to be $64 billion annually in the United States. Neurodevelopmental deficits cannot currently be reliably diagnosed until 3 to 5 years of age. Our objective is to apply deep learning to MRI acquired at term equivalent age for early prediction/investigation of later neurodevelopment deficits (cognitive, language, and motor) in very preterm infants. We analyze quantitative brain anatomical features (e.g., focal brain injury, cortical maturation metrics) and brain network connectivity features (functional and structural connectome) independently and jointly, to predict neurodevelopmental deficits, and to potentially uncover the underlying mechanisms leading to such deficits. Our models’ feature-ranking algorithms to identify predictive markers are further validated by our expert-evaluators who incorporate prior physiology and epidemiology studies ranked by evidence to iteratively refine the models and the feature-ranking. This generates a set of validated predictive brain imaging markers that are identified long before cognitive, language, or motor abnormalities arise.
Our techniques can enable the building of high-accuracy risk prediction models, whose achievements will be a milestone in bridging the translational gap and promoting improved clinical care for high-risk newborns. It will allow accurate parent counseling and early intervention therapy recommendations tailored to an individual infant’s needs. Those infants identified as being at the highest risk for adverse neurodevelopmental outcomes could then be immediately referred for more frequent surveillance and appropriateness of available interventions, thus resulting in earlier clinical allocation of emerging therapies and stimulating the design of early, targeted neuroprotective interventions. This would occur at far earlier ages, during periods of optimal neuroplasticity, than is currently possible. Those who are identified as lower risk would continue to be followed, but with lower frequency and intensity, thus decreasing health care costs and parental anxiety. Our techniques can enhance accuracy in predicting neurodevelopmental deficits for individual infants and advance personalized medicine.
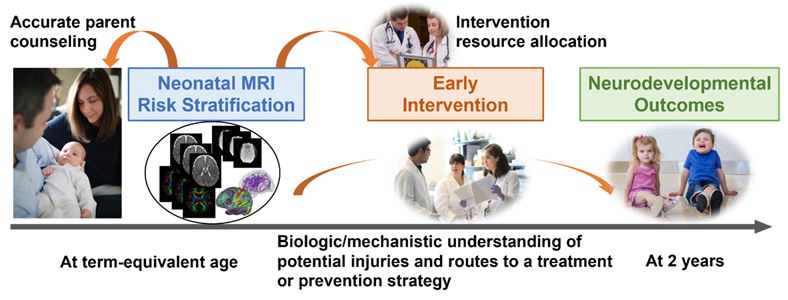
North America’s Largest Annotated Advanced MRI Very Preterm Infant Cohort.
Advanced MRI with linked clinical and outcome data are rare in very preterm infants and none exist publicly. Our two R01 projects from NINDS (2016-2021, PI, Parikh; Co-I, He) facilitated the enrollment of the population-based Cincinnati Infant Neurodevelopment Early Prediction Study (CINEPS). The CINEPS cohort has enrolled 392 very preterm infants and performed high-quality anatomical, diffusion, and functional MRI at term-equivalent age, as well as neurodevelopmental assessments at 2 years of age. Together with our previous cohort of 110 very preterm infants that have already undergone advanced MRI and 2-year neurodevelopmental assessments, we have formed North America’s largest annotated advanced MRI very preterm infant cohort. This unique large-scale cohort has established a strong foundation to develop deep learning models for early prediction of cognitive, language, and motor deficits at 2 years of age in very preterm infants.
White Matter Abnormality Quantification
Diffuse white matter abnormality (DWMA) is characterized by the diffusely higher signal intensity in white matter at T2-weighted MRI or lower signal intensity in white matter at T1-weighted MRI. It is commonly observed in very preterm infants (≤ 32 weeks gestational age) and reported in many previous studies. About 50%–80% of very preterm infants exhibit DWMA when undergoing MRI at their term-equivalent age. Despite the high prevalence of DWMA, the pathologic clinical importance of DWMA continues to be debated. Several neurodevelopmental follow-up studies have revealed that DWMA is predictive of lower developmental scores. Nevertheless, other studies have not observed substantial links between DWMA and neurodevelopmental impairments. The limited progress in resolving the controversy over DWMA is partially attributed to the lack of reproducible quantification methods for evaluating DWMA. Subjective and qualitative assessment of DWMA is still the most commonly used strategy, which may produce measurement errors, resulting in poor reliability and reproducibility. Therefore, an accurate, objective, and quantitative assessment of DWMA is essential to settle the debate of whether DWMA is a transient benign phenomenon or a pathologic abnormality. We develop deep learning models for more accurate DWMA quantification. We reported a strong association between DWMA and standardized cognitive and language scores in very preterm infants. We also externally validated that objectively quantified DWMA is an independent predictor of neurodevelopmental deficits.
Application of Deep Learning to Infant Brain MRI
Advances in MRI enable the noninvasive visualization of infants' brains through acquired multi-modal images. Researchers have shown that the quantitative MRI features may account for brain structural and functional heterogeneity in preterm infants, allowing for the identification of patients likely to develop neurodevelopmental deficits. Accurately analyzing quantitative MRI features, including anatomical and connectivity features, affords unique opportunities to study early postnatal brain development, leading to insights into the origins and abnormal developmental trajectories of neurodevelopment deficits. Through this, we may gain a better understanding of 1) how tissue characteristics and neural information are segregated and integrated; and 2) how an individual brain's organizational changes influence functions of cognition, language, and motor. Deep learning techniques are well suited for extracting biologically meaningful features and revealing discriminative information from high dimensional MRI data. The need to capture meaningful information contained in brain MRI data, together with the need to develop noninvasive prognostic tools, highlight the discipline of utilizing deep learning for MRI studies. Application of deep learning to infant brain MRI data can open up new windows into early prediction of neurodevelopmental outcomes in at-risk infants and facilitate the move towards precision medicine.
Structural and Functional Brain Connectomes: Novel Biomarkers for Neurodevelopmental Deficits
The brain connectome is a comprehensive description of the brain’s structural and functional connections in terms of brain networks. Brain connectomes form during early brain development and reshape in cases of prematurity or perinatal brain injury. The investigation of brain connectivity will improve our understanding of how an individual brain's microstructural and oscillatory neuronal activity alterations influence later cognitive, language, and motor function, resulting in improved individual risk stratification. Advances in deep learning techniques have made it possible to extract physiologically meaningful features and reveal new discriminative information from high-dimensional connectome data. There is a growing interest in developing deep learning approaches to predict adverse neurodevelopmental outcomes using brain connectivity data, but their use in the very preterm population remains relatively limited. We propose a prediction model that is capable of analyzing different types of brain connectome by fusing different neural networks via concatenation and contrastive learning. The proposed automated prognostic system uses different types of data as inputs to determine at term-equivalent age whether or not an individual very preterm infant is at high risk of developing moderate or more severe cognitive, language, and/or motor deficits, and to predict individual standardized neurodevelopmental scores (on the Bayley Scales of Infant and Toddler Development, Third Edition (Bayley III) (Bayley, 2009) Cognitive, Language, and Motor subtest scores) at 2 years corrected age.
Liver Fibrosis Prediction
Chronic liver disease (CLD) is a leading cause of morbidity and mortality in children and adults. Compared to other chronic diseases, CLD is associated with increased rates of hospitalization, longer hospital stays, and more frequent readmissions. CLD is responsible for large healthcare expenditures. A recent estimate (2017) of the lifetime costs of fatty liver disease in the U.S. was ~$222 billion. Liver fibrosis is the most important and only histologic feature known to predict outcomes from CLD, with evaluation necessary for accurate staging as well as medical and surgical decision-making. The current standard for assessing liver fibrosis is the biopsy, which is costly, prone to sampling error, and invasive with poor patient acceptance. Thus, there is an urgent unmet need for noninvasive, highly accurate, and precise diagnostic technologies for the detection and quantification of liver fibrosis. Our goal is to develop and validate clinically-effective computer-aided diagnosis techniques to help interpret liver MRI data, providing a quantitative assessment of CLD. We leverage a multi-center database of ~8,500 pediatric and adult liver MRI examinations from four institutions to develop deep learning models to identify novel imaging and clinical non-imaging biomarkers and accurately detect and quantify liver fibrosis, using biopsy-derived histologic data as the reference standard.
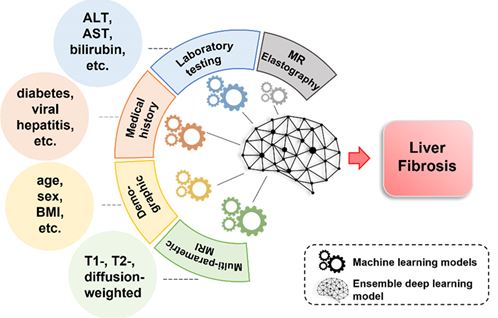
Liver Stiffness Prediction
MR Elastography (MRE) is increasingly used for detecting and assessing the severity of chronic liver disease in children and adults. MRE involves the generation of liver transverse (shear) waves using an active-passive driver system (the passive driver is placed over the right upper liver). These waves and associated displacement of liver tissue can be imaged using a modified phase-contrast pulse sequence and can be used to create quantitative images of liver stiffness, a surrogate biomarker for liver fibrosis. Although MRE obviates the need for liver biopsy in some patients and allows more frequent longitudinal monitoring of liver health, it has associated drawbacks related to additional patient time in the scanner, patient discomfort, and added costs (e.g., infrastructure and patient charge-related); the cost of adding MRE to a given MRI scanner is ~$100,000-250,000 in the U.S. for necessary hardware and software purchases. Our goal is to develop AI models to identify novel imaging and clinical non-imaging biomarkers and predict MRE-derived shear liver stiffness (~1-12 kPa). Such algorithms could theoretically direct and/or even eliminate the use of MRE, thereby decreasing imaging time and saving considerable healthcare costs (likely 10s of millions of U.S. dollars yearly).
One of the Largest Diverse Liver MRI-Pathology Datasets.
We have created one of the largest (if not the largest) multi-center (Cincinnati Children’s Hospital Medical Center [CCHMC], University of Wisconsin [UW], University of Michigan [UM], and New York University [NYU]) liver MRI-pathology datasets that is composed of both conventional anatomic and MRE images. This dataset includes ~8,500 clinical liver MRI exams from all three major manufacturers (GE Healthcare, Philips Healthcare, and Siemens Healthcare) as well as acquisitions obtained on both 1.5T and 3T clinical MR systems. Our dataset includes large numbers of scans and correlative biopsy tissue from pediatric and adult populations, as well as from patients with a variety of causes of CLD (e.g., NAFLD/non-alcoholic steatohepatitis [NASH], viral hepatitis, autoimmune liver diseases, and biliary atresia).
Histologic Liver Fibrosis Assessment.
Available tissue specimens in the form of existing stained slides (including Masson trichrome or Sirius red stained), recut unstained slides, and/or paraffin blocks have been obtained. All recut unstained slides were undergone staining at CCHMC as a batch using a fibrosis-specific stain (e.g., Masson’s trichrome). At least two slides from each subject were reviewed separately and scored for the presence and amount of fibrosis by two study expert hepatopathologists using a validated semi-quantitative staging system (e.g., METAVIR). Slides also were undergone digital scanning and the fibrosis percentage (0-100%) on each slide was quantified as measured by the collagen proportionate area using an existing computer-based algorithm; two slides were scanned per subject with the fibrosis percentage averaged.
Liver and Spleen Segmentation
Automated and accurate liver and spleen segmentation is crucial to extract radiomic and deep features, but it remains challenging due to high inter-subject variability in organ size, shape, signal intensity/appearance, and close proximity to other organs. Previous efforts have been made to perform automated abdominal organ segmentations on computed tomography and MRI. However, most of the existing methods only produce moderate accuracies for a single image type (e.g., T1- or T2-weighted images). There has been a general lack of application to multiparametric MRI data. To improve the segmentation performance, we propose to develop a method that simultaneously uses multiparametric MRI data (e.g., T1-, T2-, and diffusion-weighted images of the abdomen) as inputs and jointly segments the liver and spleen.
Grants / funding:
NIH, R01 EB029944
NIH, R01 NS094200
NIH, R01 NS096037
NIH, R21 HD094085
NIH, R01 EB030582



